Etiquetas
Dimitriv Ivanovich Mendeliév a Russian who 150 years ago presented a periodic table to gather all the elements including those that were to be discovered.
The United Nations has declared 2019 as the International Year of the Periodic Table of Chemical Elements (IYPT2019). In the mid-nineteenth century 69 elements were known but they were not organized. In 1860, if he fixed the concept of «atomic weight» in the International Congress of Chemists in Kalrsruhe (Germany), Mendeliév presented his periodic table in 1869: he ordered the elements according to their atomic weight, marking the periodic tendencies and the elements with similar behavior in the same column.
Tool of the chemical elements of the Earth and the rest of the universe, used in chemistry, physics and biology.
Dimitri Mendeliév is often referred to as the “Father of the Period Table”, he called his table “The Periodic System”.
Mendeliév made important contributions to chemistry and other fields: hydrodynamics, meteorology, geology and branches of chemical technology. Was one of founders, in 1869, of the Russian Chemical Society.
Dmitri Ivanovich Mendeleev (1834-1907) was a Russian chemist and inventor. Mendeliév became a professor at the Saint Petersburg Technological Institute (while he was teaching at various schools, he completed his first chemistry textbook “Organic Chemistry”) and Saint Petersburg State University in 1864. In 1867 he started to teach inorganic chemistry.
1861 constituted a period of great change and reform in Russia: social and economic change after the social and political reforms of Peter the Great. “The Great Reforms Era”, it was also a time of change in chemistry: the emergence of a new generation of chemists in Russia. The Russian Chemical Society was organized in 1868, Mendeliév being one of the founding members. The objectives that Russian chemists was to educate qualified professionals for the new capitalistic production that Russian required. The classical foundations of chemistry were being established.
Mendeliév found that the chemical properties of elements are a function of their atomic weights. The atomic number is the number of protons that an atom contains and has an important influence in the arrangement of electrons that define the chemical properties of the element.
In 1863 there were 56 known elements. An element occupies a specific place in the period system. He found that the chemical properties of elements are a function of their atomic weights. He arrived at his discovery through a search for the “grammar” of the elements (using the “isomorphism” and looking for general formulas to generate the possible chemical compounds. Arranged the elements in order of increasing atomic weights, and called the first row (oxygen, nitrogen, carbon etc.) “typical elements”. His inorganic chemistry textbook “The Principles of Chemistry” an inorganic textbook.
His doctoral thesis “On Compounds of Alcohol with Water” submitted in 1865. Mendeliév was moving away from a belief in the atomic theory in this period (1864-1868).
“The Nature of the Chemical Elements as a Function of their Atomic Weights”, appeared early in 1870. He began with speculation related to Prout´s hypothesis.
By adding additional elements following this pattern, he developed his extended version of the periodic table. On 6 March 1869, he made a formal presentation to the Russian Chemical Society, titled “The Dependence between the Properties of the Atomic Weights of the Elements”, which described elements according to both atomic weight and valence. This presentation stated that:
1The elements according to their atomic weight, exhibit an apparent periodicity of properties.
2. Elements which are similar regarding their chemical properties have similar atomic weights.
3. The elements in groups of elements in the order of their atomic weights corresponds to their so-called valencies.
4. The elements which are the most widely diffused have small atomic weights.
5. The magnitude of the atomic weight determines the character of the element.
6. We must expect the discovery of many yet unknown elements.
7. The atomic weight of an element may sometimes be amended by a knowledge of those of its contiguous elements.
8. Certain characteristic properties of elements can be foretold from their atomic weights.

He was elected a Foreign Member of the Royal Society of London (ForMemRs) in 1982 and in 1983 he was appointed director of the Bureau of Weights and Measures, a post which he occupied until his death. He also investigated the composition of petroleum and is given credit for the introduction of the metric system to the Russian Empire.
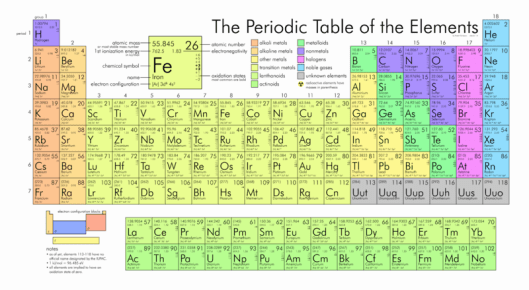
The periodic Table: families and periods
In the periodic table of elements, there are seven horizontal rows of elements called periods. The vertical columns of elements are called groups, or families.
Periods in the periodic table
In each period (horizontal row), the atomic numbers increase from left to right. The periods are numbered 1 through 7 on the left-hand side of the table.
Elements that are in the same period have chemical properties that are not all that similar.
Families in the periodic table
Members of the families (vertical columns) in the periodic table have similar properties. The families are labeled at the top of the columns in one of two ways:
• The other method uses Roman numerals and letters.
• The newer method uses the numbers 1 through 18.
The figure below lists some important families that are given special names:
• The IA family is made up of the ALKALI METALS
• The VIIA family is made up of the HALOGENS
• The VIIIA family is made up of the NOBLE GASES
Valence electrons and families
An electron configuration shows the number of electrons in each orbital in a particular atom. These electron configurations show that there are some similarities among each group of elements in terms of their valence electrons.
Bibliography:
• Masanori Kaji; “D.I. Mendeliev´s concept of chemical elements and the principles of chemistry”; Bull. Hist. Chem.; Tokyo Institute of Technology; 2002
• Dorling Kindersley; “The Periodic Table Book” a Visual Encyclopedia of the Elements; Dorling Kindersley Publishing; 2017
• R.J. Puddephatt; P.K. Monaghan; “The Periodic Table of Elements”, Oxford Chemistry Series; 1986
• “Mendeléeff, Dmtri Ivanovich”, Encyclopedia Britannica. Cambridge University Press; 1911
https://en.wikisource.org/wiki/1911_Encyclop%C3%A6dia_Britannica/Mendel%C3%A9eff,_Dmitri_Ivanovich
Relationated links:
• UNESCO: events
https://www.iypt2019.org/events
• Periodic tablet app
http://www.rsc.org/periodic-table
• Mendeliév y la tabla periódica. Alexander Rojas
